Original article (in Serbian) was published on 11/05/2022
A post with dramatic “new Pfizer data” was published on the Viber group “Doctors and parents for science and ethics”, which is known for its anti-vaccine attitudes and promotion of ivermectin. A member of the group claims that out of 270 pregnant women who were monitored by this company after receiving the vaccine, 238 “left the study for an unknown reason”, and the remaining pregnant women, except for one, gave birth to dead children or had miscarriages. To make it seem more convincing, this member also attached one page from Pfizer’s research in English to the claims, and he additionally “spiced up” the drama using a picture of a syringe and a coffin. However, these are all manipulatively interpreted data.
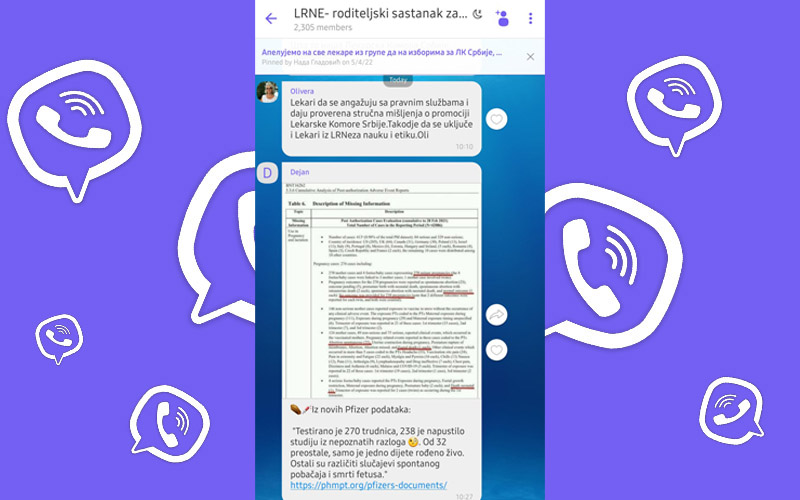
“From the new Pfizer data: “270 pregnant women were tested, 238 left the study for unknown reasons. Of the 32 remaining, only one child was born alive. There are still various cases of miscarriage and fetal death”, Dejan wrote, providing a link to the database of Pfizer’s documents and a photo from the research he is referring to, where these numbers are underlined in red. He put these claims in quotation marks to make them look like a quote from a document.
The document does exist, but the numbers have been interpreted incorrectly, apparently to cause panic and deter pregnant women from vaccination.
What is it all about?
The data referred to by the member of this Viber group is not “new” – it is a document from 2021 featuring statistics from 63 countries regarding side effects of the vaccine, which were reported from December 1, 2020, to February 28, 2021.
The section on vaccinated pregnant women (on page 12) contains a table stating that 270 of them were monitored during this period. In 23 cases, meanwhile, there was a miscarriage, in two cases, the fetus died, while in one case, the baby died soon after birth, and only one baby was born without any problems.
These numbers, however, do not prove that all these things, which happened after getting the vaccine, also happened because of the vaccine.
As the study states at the outset, all adverse events reported – in pregnant women and all other vulnerable categories covered by the report – do not necessarily have to do with the vaccine.
“The number of adverse event reports does not necessarily mean that a particular adverse event is caused by a drug (vaccine), it may be caused by an underlying disease or other factors such as a medical history or a drug being taken at the same time”, the report stated.
The remaining 238 pregnant women are said to have “no outcome”, which a member of this Viber group interprets as “they left the study for an unknown reason”.
The Reuters fact-checking team also dealt with this disinformation in January this year. As Emily Adhikari from the Department of Maternal and Fetal Medicine at the University of Texas told the agency, it is wrong to assume that 238 cases with “no outcome” at the time of this study ended in a negative outcome.
In other words, Reuters states, “no outcome” only means that those pregnancies were in progress at the time of this report.
Besides the fact that based on this data from a year and a half ago, it cannot be concluded that spontaneous abortions and fetal deaths necessarily occurred due to the vaccine, it cannot be concluded that all pregnant women who receive the Pfizer vaccine are endangered.
Namely, the report covers 270 vaccinated pregnant women, which is only a small percentage of the total number. According to the American Center for Disease Control and Prevention data, by February 16, 2021 (the period covered by the report), over 30,000 pregnant women were vaccinated in the United States alone.
When this data was collected, Pfizer’s BioNTech vaccine was approved by the US Food and Drug Administration only for emergency use in the most endangered patients.
The document in question was submitted to the Administration in 2021 and is one of the numerous reports that Pfizer submitted last year in the process of registering a vaccine that would be used not only in emergencies but would be registered and approved for the general population.
In the meantime, the course of vaccination and adverse events after receiving the vaccine were monitored, so BioNTech was registered in the United States in August 2021 for the widest population because it was estimated to be sufficiently effective and safe. According to the Food and Drug Administration website, pregnant women can also use it in consultation with doctors.
“There are no contraindications for receiving the vaccine in pregnant or breastfeeding women. “Pregnant or breastfeeding women should discuss the potential benefits and risks of vaccination with their doctor”, the Administration writes.
The American Center for Disease Control and Prevention believes that there is a reason for optimism when it comes to vaccinating pregnant women. Data from their national database of adverse events during pregnancy after vaccination have so far not shown major reasons for concern for the health of pregnant women or babies. In addition, as they point out, several large studies so far have shown no increased risk of miscarriage or premature birth in women who have received the vaccine.
Even after the vaccine was registered in the United States, Pfizer continued its studies on the vaccine’s safety in pregnant women, so the data is still being collected.
The European Medical Agency said in January this year that it had analyzed data on 65,000 monitored pregnancies in women who received the vaccine – mostly EU-approved Pfizer or Moderna vaccines. They said they did not find any signs of an increased risk of complications in pregnancy, miscarriage, premature birth or side effects in unborn babies due to vaccination.
“Given that pregnancy has so far been associated with an increased risk of developing severe COVID-19, especially in the second and third trimesters of pregnancy, women who are pregnant or could become pregnant in the near future are encouraged to be vaccinated in accordance with national recommendations”.
In Serbia, the vaccine developed by this company is given to pregnant women, with a note that it is necessary to consult a doctor beforehand.